TCAR procedure offers potentially safer method for high-risk stroke patients
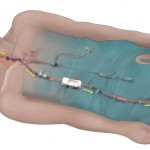
[News-Medical.net, January 18, 2016] The FDA recently approved a new TransCarotid Artery Revascularization (TCAR) procedure that utilizes the ENROUTE® Transcarotid Neuroprotection and Stent System from Silk Road Medical. The TCAR procedure offers patients a potentially safer method of carotid stenting through a small cut down at the base of the neck and direct carotid artery access along with neuro-protective flow reversal during delivery of the stent.
The Minneapolis Heart Institute Foundation® (MHIF) was selected as the only center in the four state area to participate in the ROADSTER 2 Study. Jessica Titus, MD, and Timothy Sullivan, MD, vascular surgeons at Minneapolis Heart Institute® at Abbott Northwestern Hospital, will serve as Principal Investigator and Co-Investigator, respectively, for the trial.
"We are excited to be able to bring this carotid artery revascularization option to our patients. It combines the advantages of a minimally invasive approach for high risk patients with a lower stroke risk than the standard method for carotid stent placement." said Dr. Titus. Read more at news-medical.net